
physical chemistry - Is the compressibility factor smaller or
The compressibility factor of a gas is defined as $Z = pV/(nRT)$. If attractive intermolecular forces dominate then $Z$ tends to be smaller than 1, and vice versa if repulsive forces dominate. In

Van der Waals Equation, Definition & Examples - Lesson
11.3: Critical Phenomena - Chemistry LibreTexts

Compressibility factor - Wikipedia
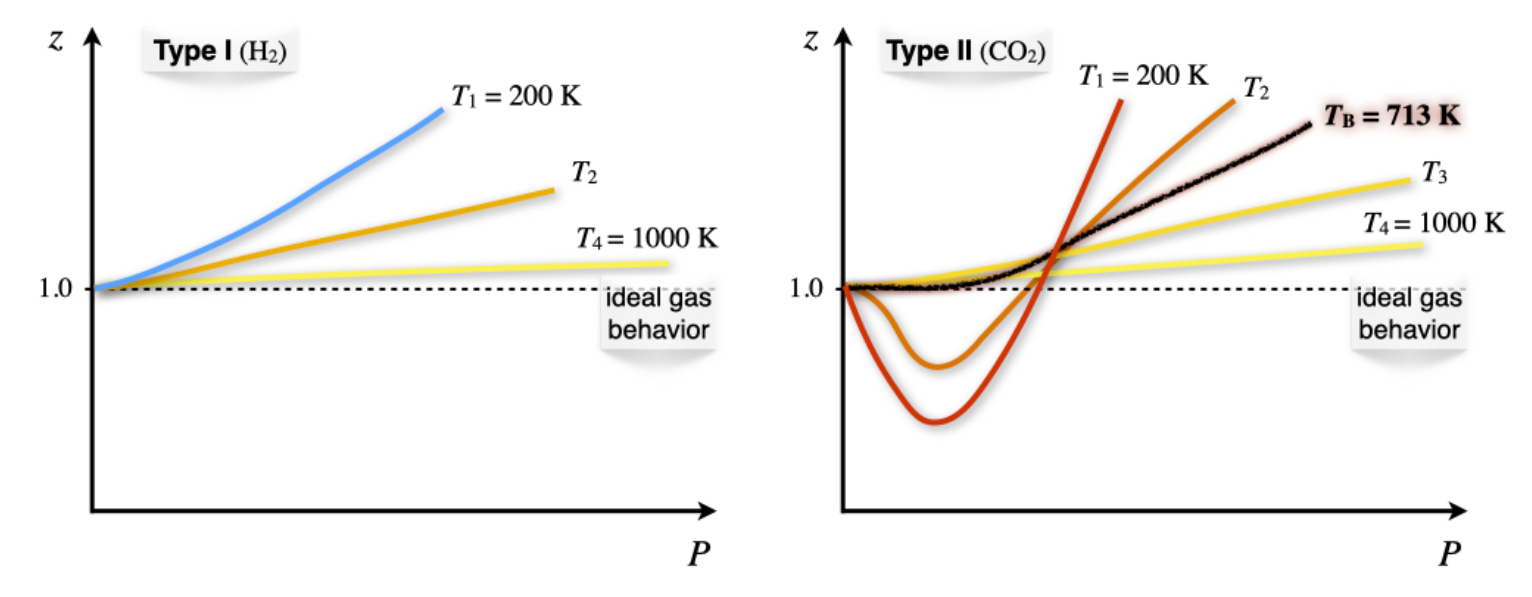
11.3: Critical Phenomena - Chemistry LibreTexts

Non-Ideal Gas Behavior Chemistry: Atoms First

Gas compressibility factor Z: Ideal gas vs Real gas

Compressibility factor (gases) - Citizendium

Other Causes of Limb Ulcers Causes Physical or chemical injury

Compressibility factor (gases) - Knowino
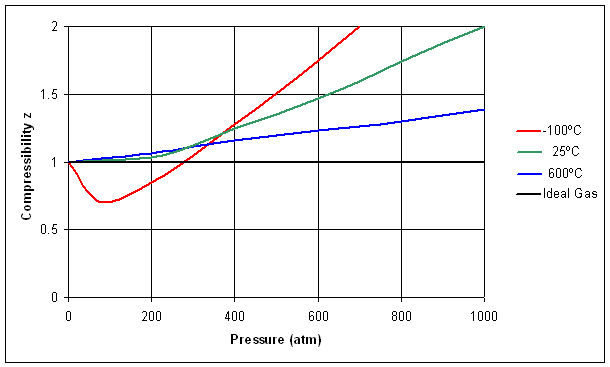
Gas Laws – First Year General Chemistry

Experiments. - ppt video online download

Physical Chemistry The Compression Factor (Z) [w/1 example

Cubic equations of state - Wikipedia

Physical Chemistry The Compression Factor (Z) [w/1 example]

Compressibility of Liquids - an overview









