
the equation of state of a gas is p(v-nb)=rt where b and r are consta - askIITians
the equation of state of a gas is p(v-nb)=rt where b and r are constants. if the pressure and temperature are such that vm=10b what is the value of compressibi

Solved PV RT The ideal gas equation states that = n, where P

The equation of state of some gases can be expressed as (P+ 2 (v - b) = RT , where P is the pressure, V is the volume, T is the absolute

Solved Q4/ The ideal gas equation of states is given by: PV

⏩SOLVED:12-104 For a gas whose equation of state is P(v-b)=R T the…
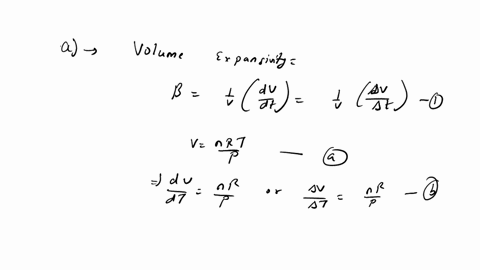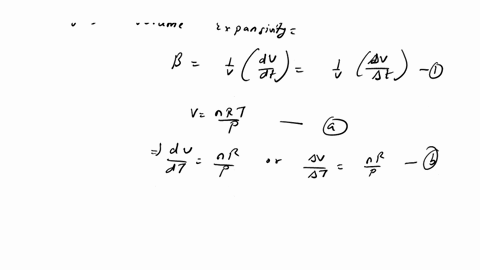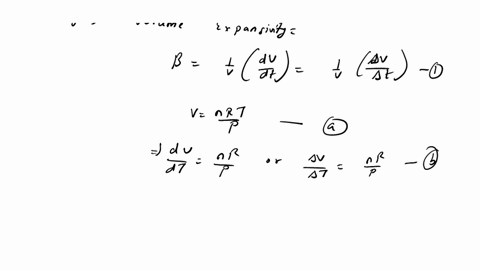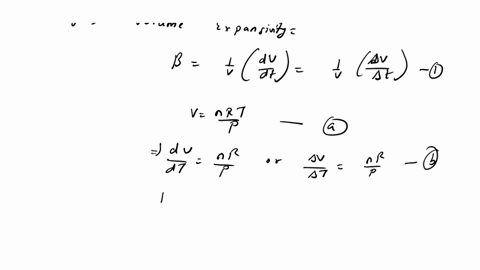
⏩SOLVED:The equation of state of an ideal gas is P V=n R T, where n…


The equation of state of a real gas is given by `(P+a/V^(2)) (V-b)=RT` where P, V and T are pre

The equation of state for a gas is P(V−nb)=nRT, where b &R are constant. ..
How is the Combined Gas Law derived? - Quora

⏩SOLVED:A gas obeys the equation p(V-b)=R T where b is a constant.…

Solved The van der Waals equation of state is given as (P +

Deviation From Ideal Gas Behavior - Study Material for IIT JEE
How is the Combined Gas Law derived? - Quora

the equation of state of a gas is p(v-nb)=rt where b and r are consta - askIITians







