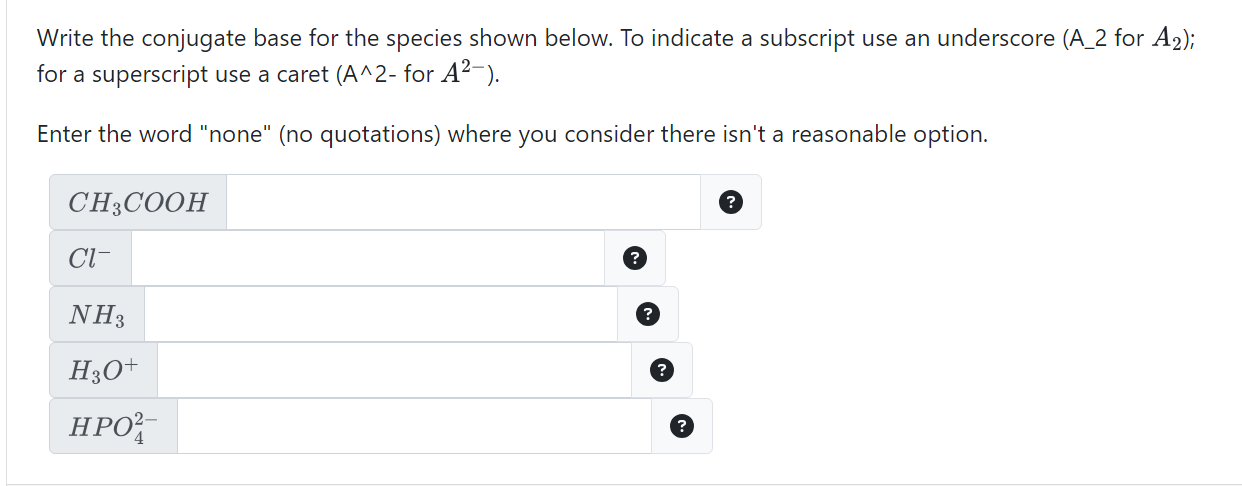
Solved Write the conjugate base for the species shown below.

For the following reaction, identify the reactant that is an acid, the reactant that is a base, and the two conjugate acid-base pairs present. OH-(aq) + HNO2(aq) arrow H2O(l) + NO2-(aq)

Solved 51. Label the acid, base, conjugate acid, and

Acid and Base Strength

In the Acid-Base reaction shown below, write the structure of the conjugate base of tropolone. Using curved arrow notation write down all possible resonance structures for this conjugate base.
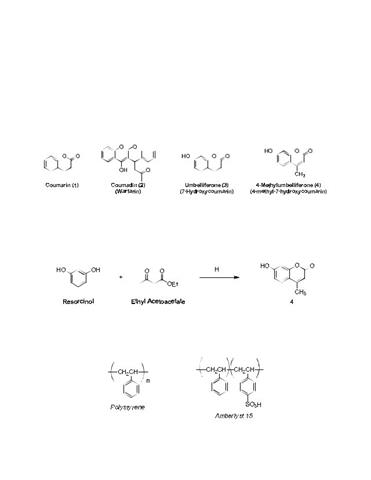
Solved) - (Part of post lab questions 1, 2, and 9) I need help drawing all (1 Answer)

Titration curves & equivalence point (article)

Solved 3. In the equilibrium shown below, which species is
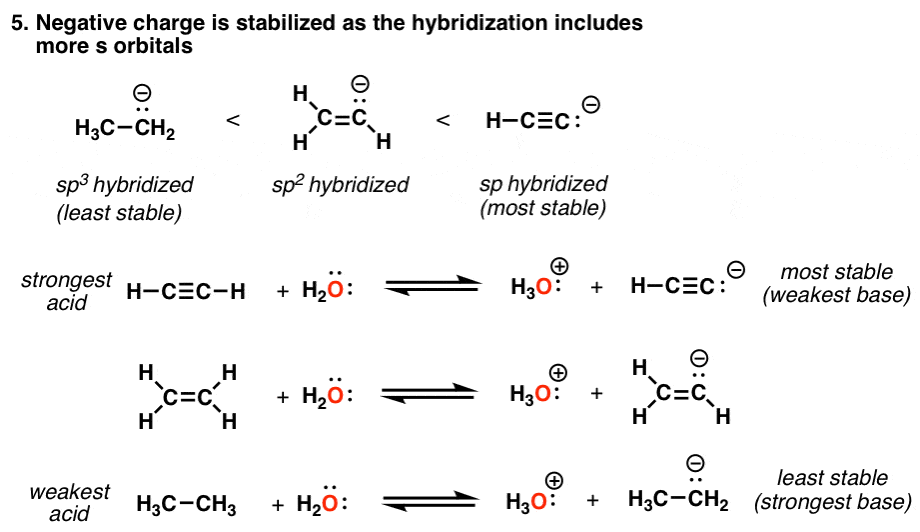
Acidity Trends In Organic Chemistry – Master Organic Chemistry
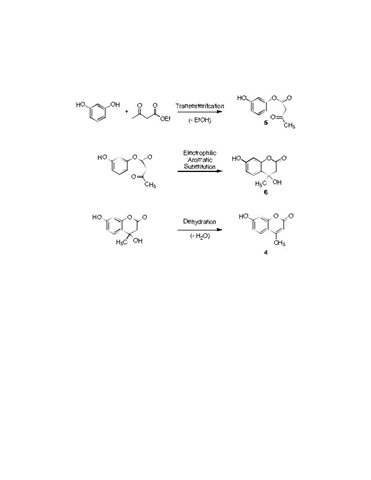
Solved) - (Part of post lab questions 1, 2, and 9) I need help drawing all (1 Answer)
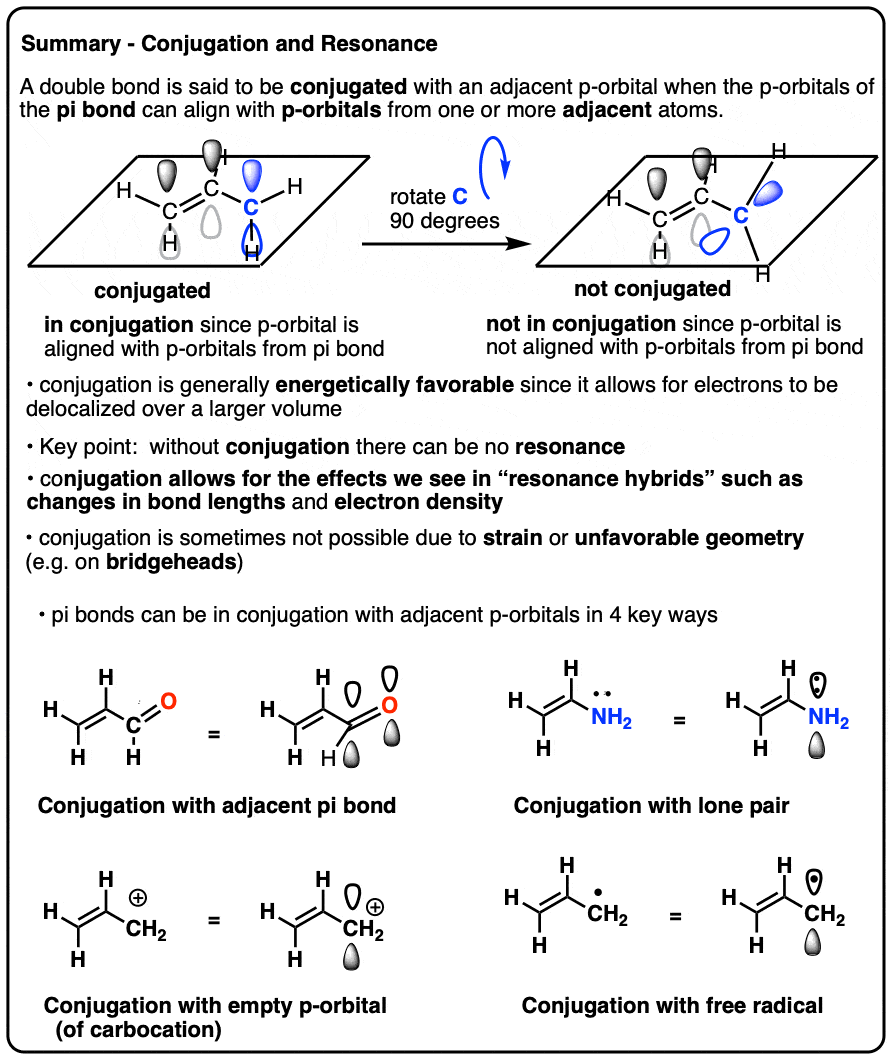
Conjugation And Resonance In Organic Chemistry

Write the reaction of glycine acting as a base. Show the structure of glycine under physiological conditions. Include charges. Omit lone pairs and explicit hydrogens.

Answered: 1. What species is the conjugate acid…
/chapter3/pages33and34/page33and34_files/aqh3o.png)
Chapter 3
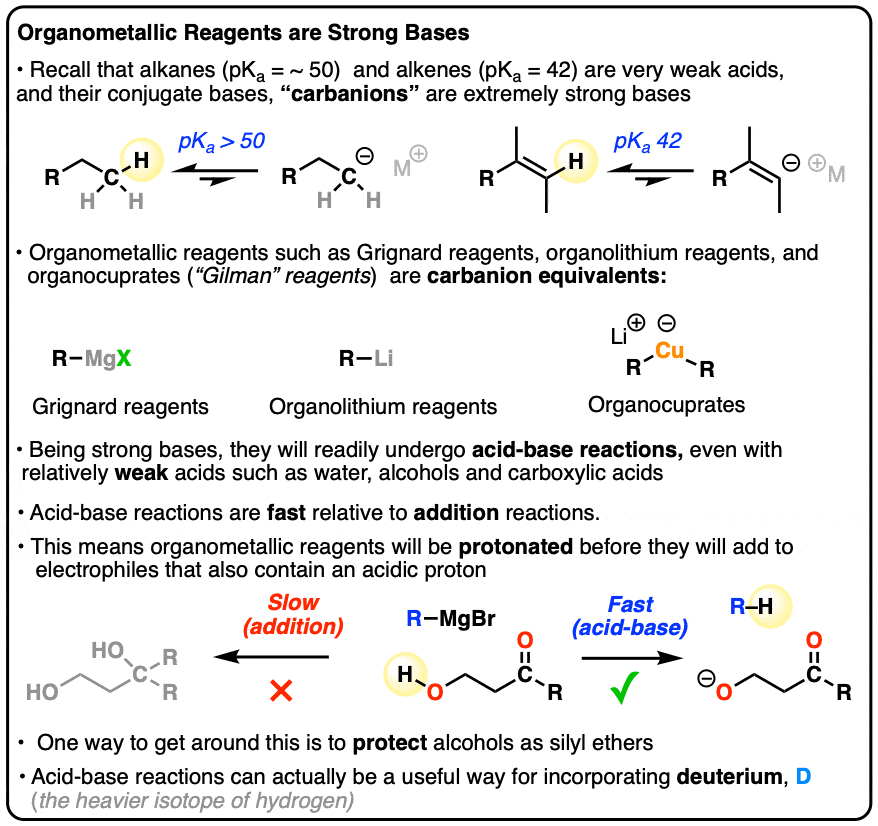
Why Grignard Reagents React With Water – Master Organic Chemistry









