Shapes of Atomic Orbitals - Shape of s, p, d, f Orbitals, FAQs, Examples
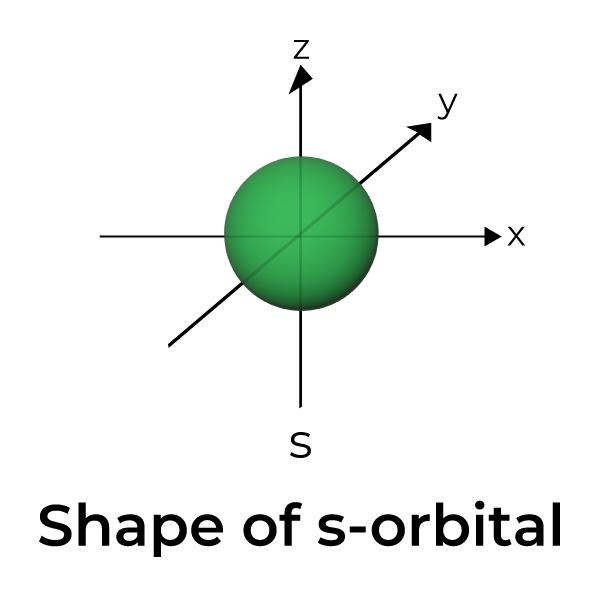
Atomic Orbitals are the three-dimensional space near the nucleus of an atom where the possibility of discovering an electron is maximum. The shape of an atomic orbital is associated with the quantum number and the angular momentum of the atom. Different types of atomic orbitals are. s-orbitals , p orbitals, d-orbitals and f-orbitals.
A computer science portal for geeks. It contains well written, well thought and well explained computer science and programming articles, quizzes and practice/competitive programming/company interview Questions.
Atomic orbitals, which are mathematical functions, describe the wave behavior of electrons (or electron pairs) in an atom. Atomic orbitals have different shapes according to their type. The different shapes of atomic orbitals describe that there is either the highest or zero probability of finding an electron around the nucleus
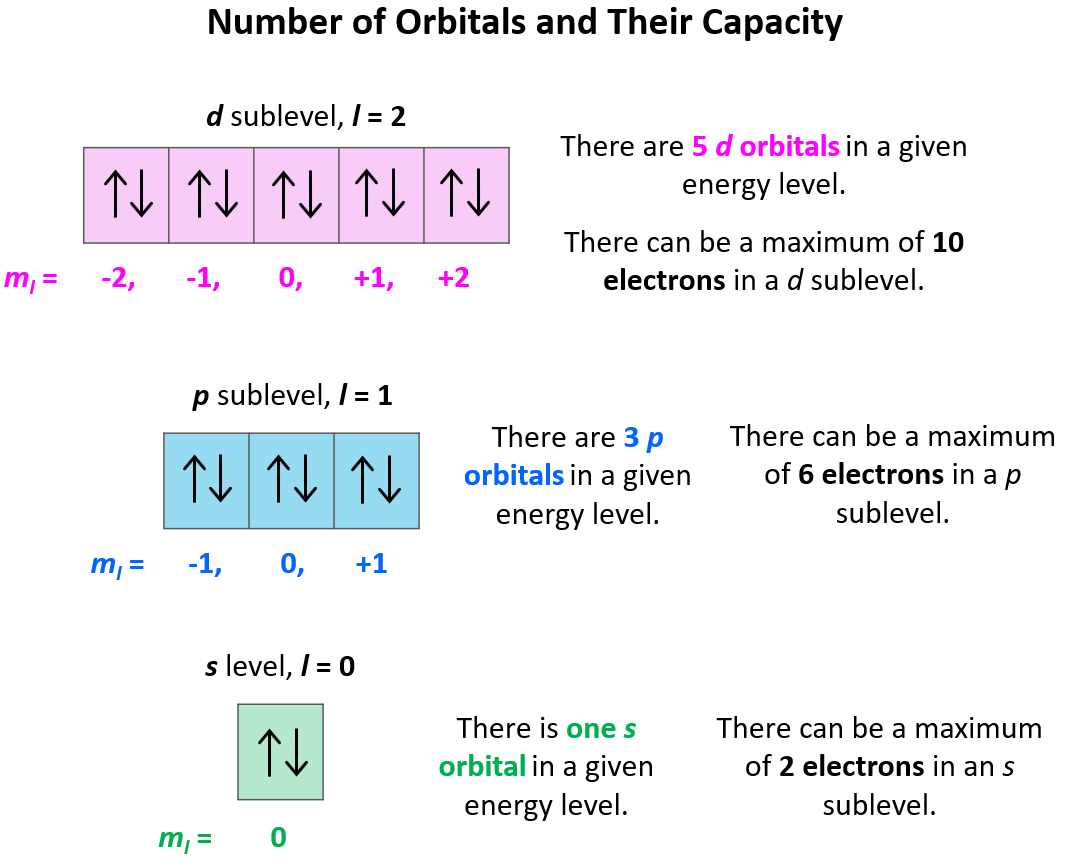
s, p, d, f Atomic Orbitals - Chemistry Steps
shapes of orbitals
Why are p, d, and f orbitals not symmetric about the rotation
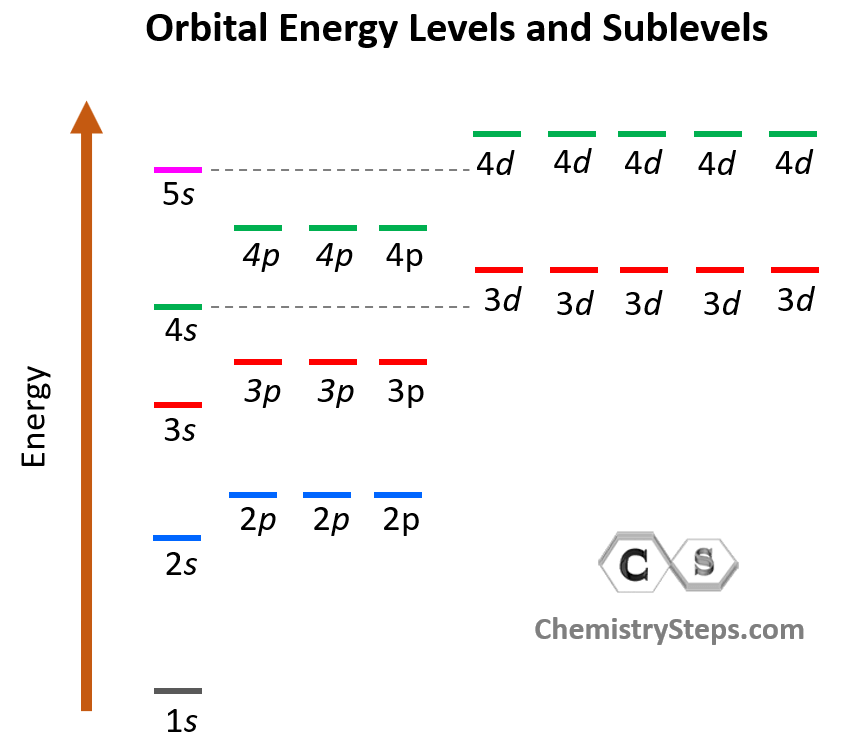
s, p, d, f Atomic Orbitals - Chemistry Steps
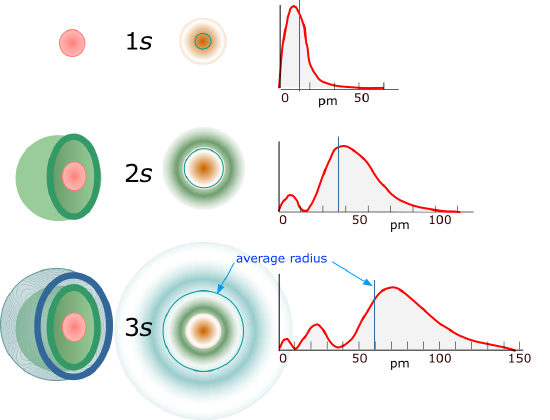
6.6: The Shapes of Atomic Orbitals - Chemistry LibreTexts

Telugu] Define atomic orbital . Explain the shapes of s, p and d orbi

Atomic Structure: Discoveries, Models, and Subatomic Particles

Atomic orbital - Wikipedia

Electron Orbitals Definition, Subshells & Shapes - Lesson

Shapes of Atomic Orbitals — Overview & Examples - Expii

Shapes of Atomic Orbitals: Shape Determination - SPD, Videos, Examples