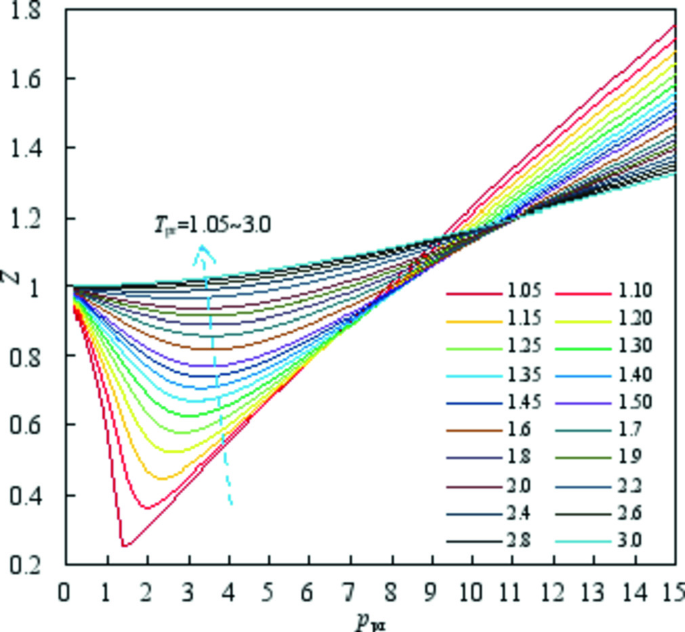
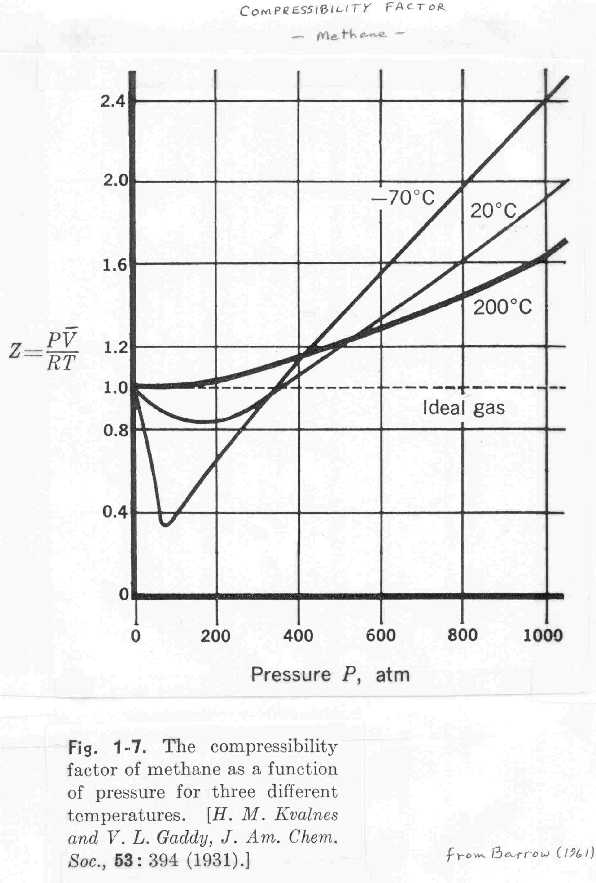
thermodynamics - Variation of compressiblity factor with
While I certainly understand the order of temperatures, I can't find a reason for the curves to intersect at one common point. Why do the curves intersect at one point? or do they really intersect
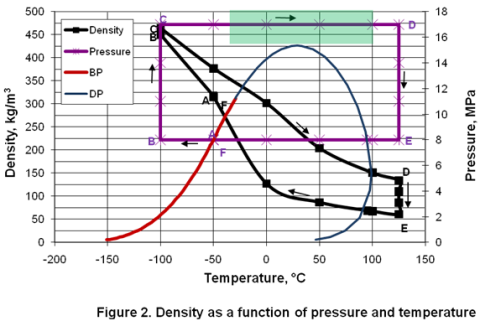
Variation of properties in the dense phase region; Part 2 – Natural Gas

Compressibility Chart - an overview

Deviation of Real Gases from Ideal Gas Behaviour - GeeksforGeeks

Energies, Free Full-Text

Compressibility factor z versus 100/V, for several values of Pressure

Compressibility factor Z as function of temperature T with lines of

The given graph represents the variations of compressibility factor `Z=PV//nRT` vs `
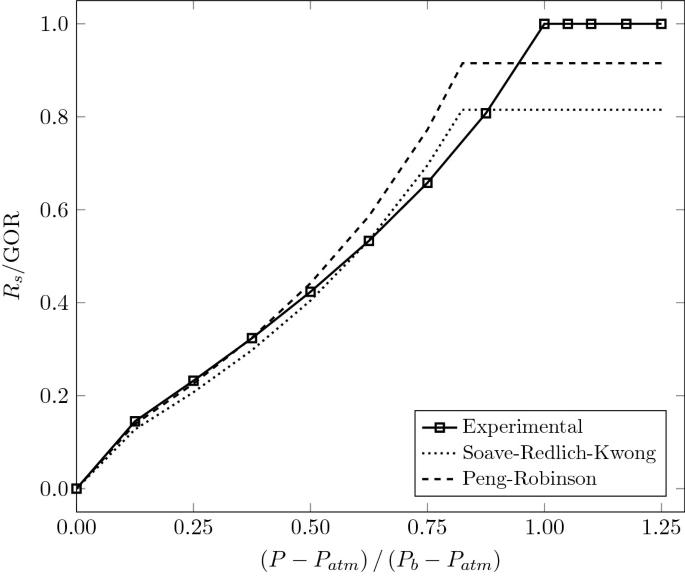
Thermodynamics of Petroleum Mixtures

Variation of internal energy (E) with the hHorizon radius
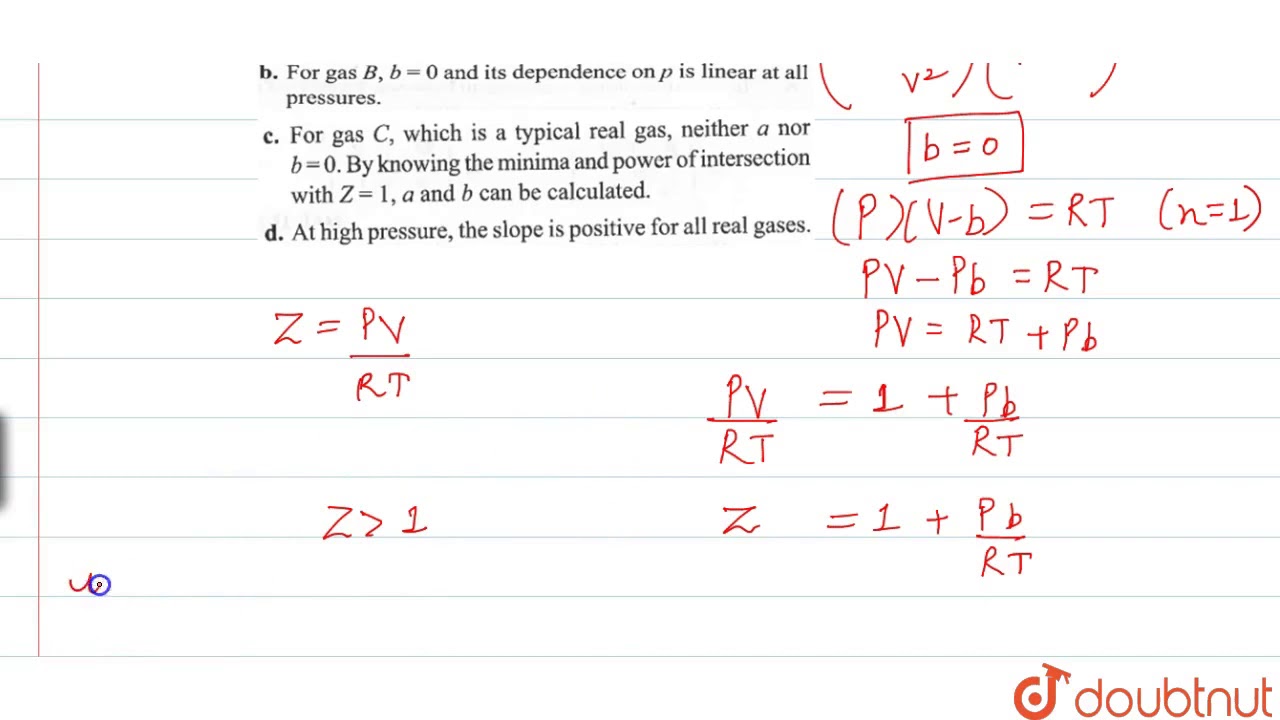
The given graph represents the variations of compressibility

The given graph represents the variations of compressibility factor `Z=PV//nRT` vs `

Compressibility factor (gases) - Citizendium