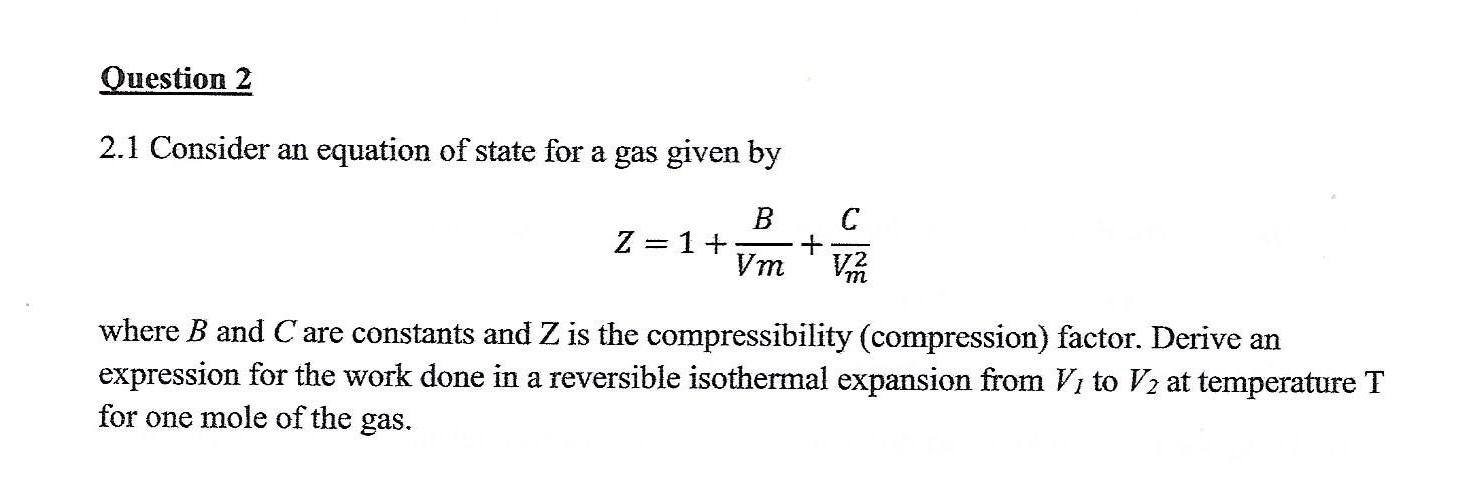Solved 9 Compression factor Z Use the van-der-Waals equation
5
(357)
Write Review
More
$ 9.50
In stock
Description

Solved We begin by showing that the compressibility factor

At low pressures, the van der Waals equation is written as [P+(a)/(V^(

Chem II - Real Gases: Van der Waals (Liquids and Solids)

Chapter 1. Properties of Gases - ppt download

Compressibility Chart - an overview

Assertion is true but Reason is false.

Original, PDF, Gases

Answered: Quantities such as the isothermal…

s3-us-west-2.aws.com/courses-images-archive

Compressibility Factor Z Important Concepts and Tips for JEE Main

Non-ideal behavior of gases (article)

Compressibility Factor of Gas Overview, Equation & Chart
Related products