Dr Alfred Msezane - Negative Ion Formation in Complex Heavy Systems •
When an electron is absorbed by a heavy atom or molecule, a heavy, negatively-charged ion is formed. These negative ions can be used for a wide array of useful applications, from organic solar cells to water purification. However, the electron absorption process for heavy particles is a complex many-body process, making it difficult for physicists to understand how the ions form. Dr Alfred Msezane at Clark Atlanta University has developed a robust mathematical theory to gain a fundamental understanding of negative ion formation in such heavy systems for the first time.
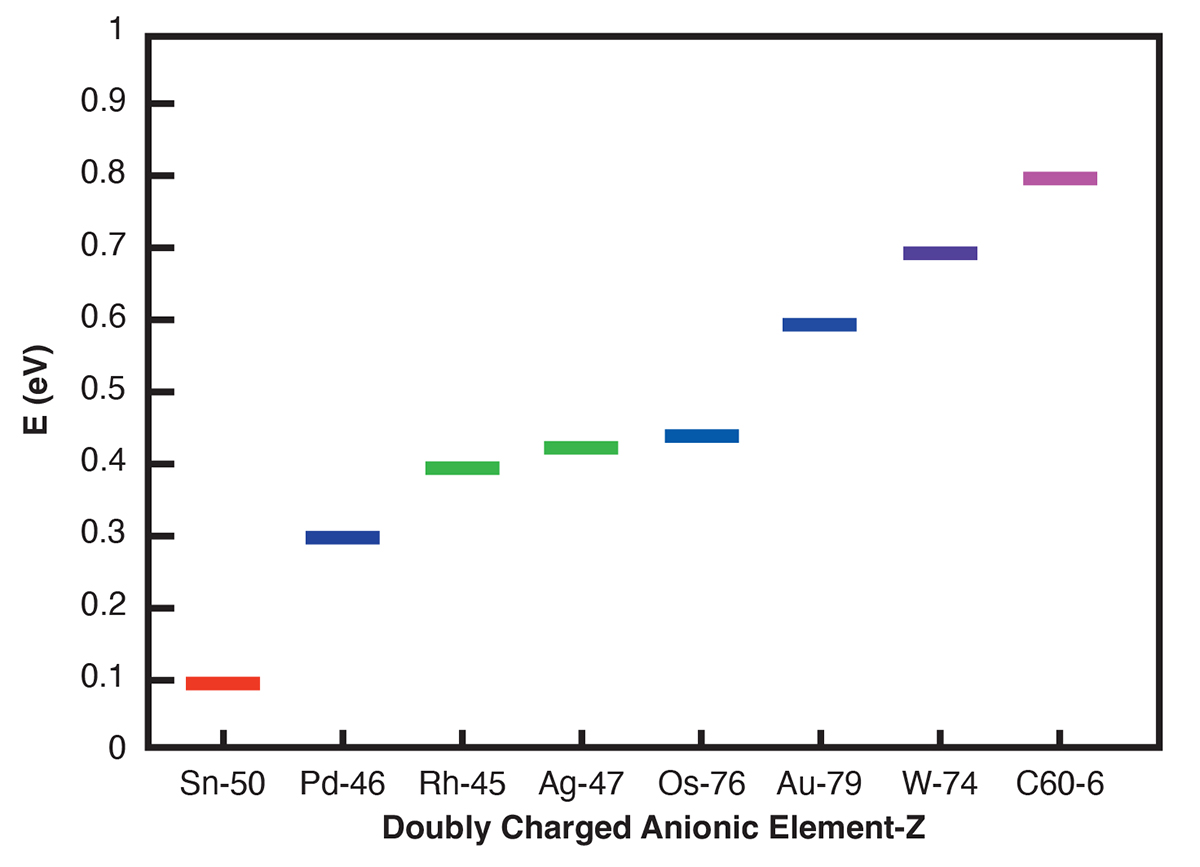
Doubly charged atomic negative ions for efficient tunable water oxidation to hydrogen peroxide

Negative Ion Formation in Complex Heavy Systems - Dr Alfred Msezane, Clark Atlanta University •

PDF) Electron Collisions with Multielectron Atoms and Fullerene Molecules: Strong Polarisation Effects

Alfred Z MSEZANE, Professor of Physics, Clark Atlanta University, Atlanta, CAU, Department of Physics
Earth & Environment Test •

Alfred Z MSEZANE, Professor of Physics, Clark Atlanta University, Atlanta, CAU, Department of Physics

PDF) Rigorous Negative Ion Binding Energies in Low-Energy Electron Elastic Collisions with Heavy Multi-Electron Atoms and Fullerene Molecules: Validation of Electron Affinities

Atoms, Free Full-Text

Alfred Z MSEZANE, Professor of Physics, Clark Atlanta University, Atlanta, CAU, Department of Physics
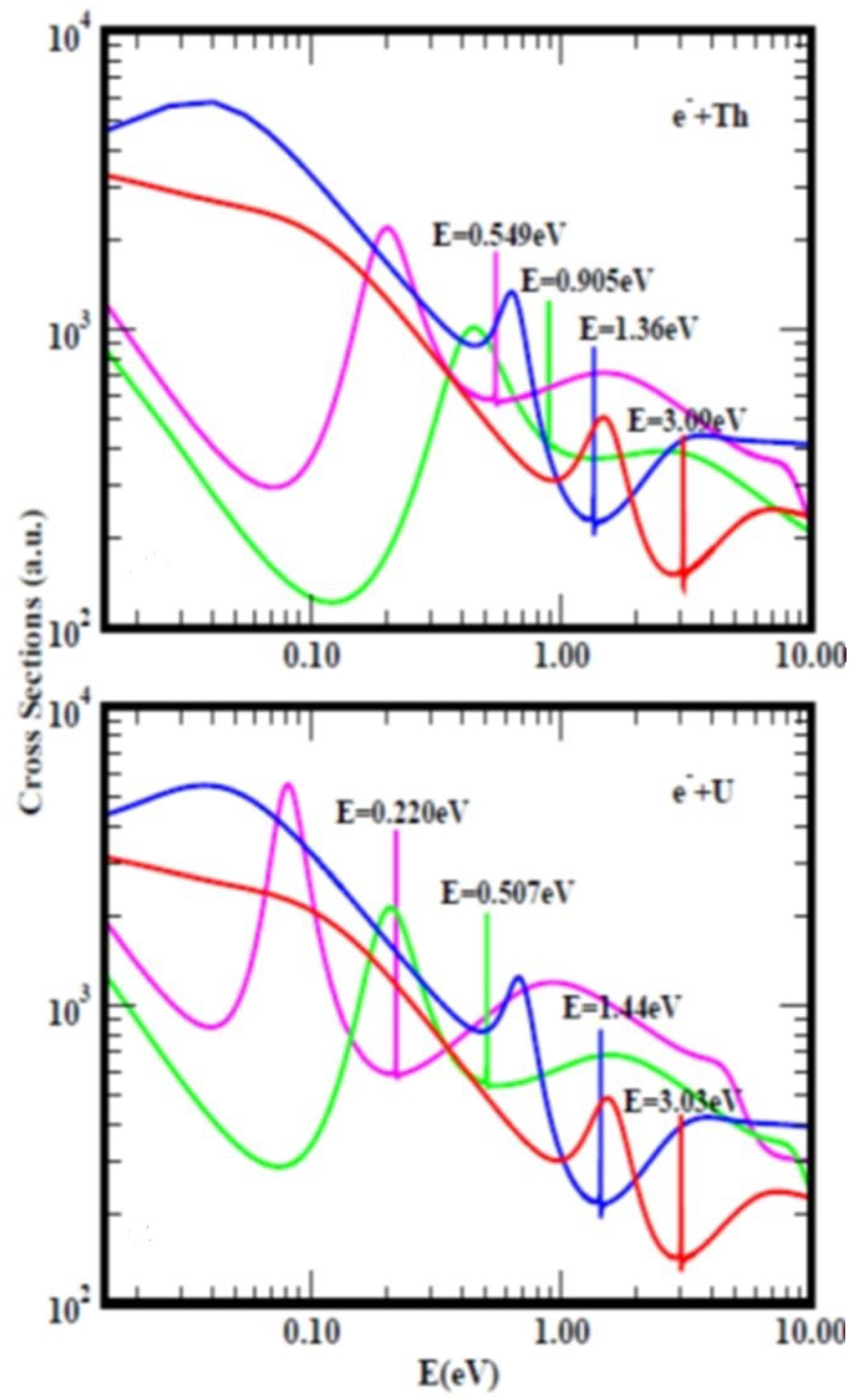
Electron Elastic-Collisions with Multi-Electron Atoms and Fullerene Molecules
Physical Sciences & Mathematics •

PDF) Negative Ion Binding Energies in Complex Heavy Systems

Alfred Msezane - Wikipedia