Antimicrobial Resistance and Medical Devices
4.6
(67)
Write Review
More
$ 27.99
In stock
Description
Antimicrobial susceptibility tests (ASTs) are medical devices used in clinical laboratories to determine which antibiotics (or other antimicrobial agents) are likely to be effective. All AST devices sold in the U.S. for use in clinical laboratories must be reviewed and cleared by the FDA’s CDRH prio

Antibiotic Resistance: A Pressing Public Health Issue - NFID

cdn.unenvironment.org/s3fs-public/inline-images/pr

Preparing for Medical Device Audits
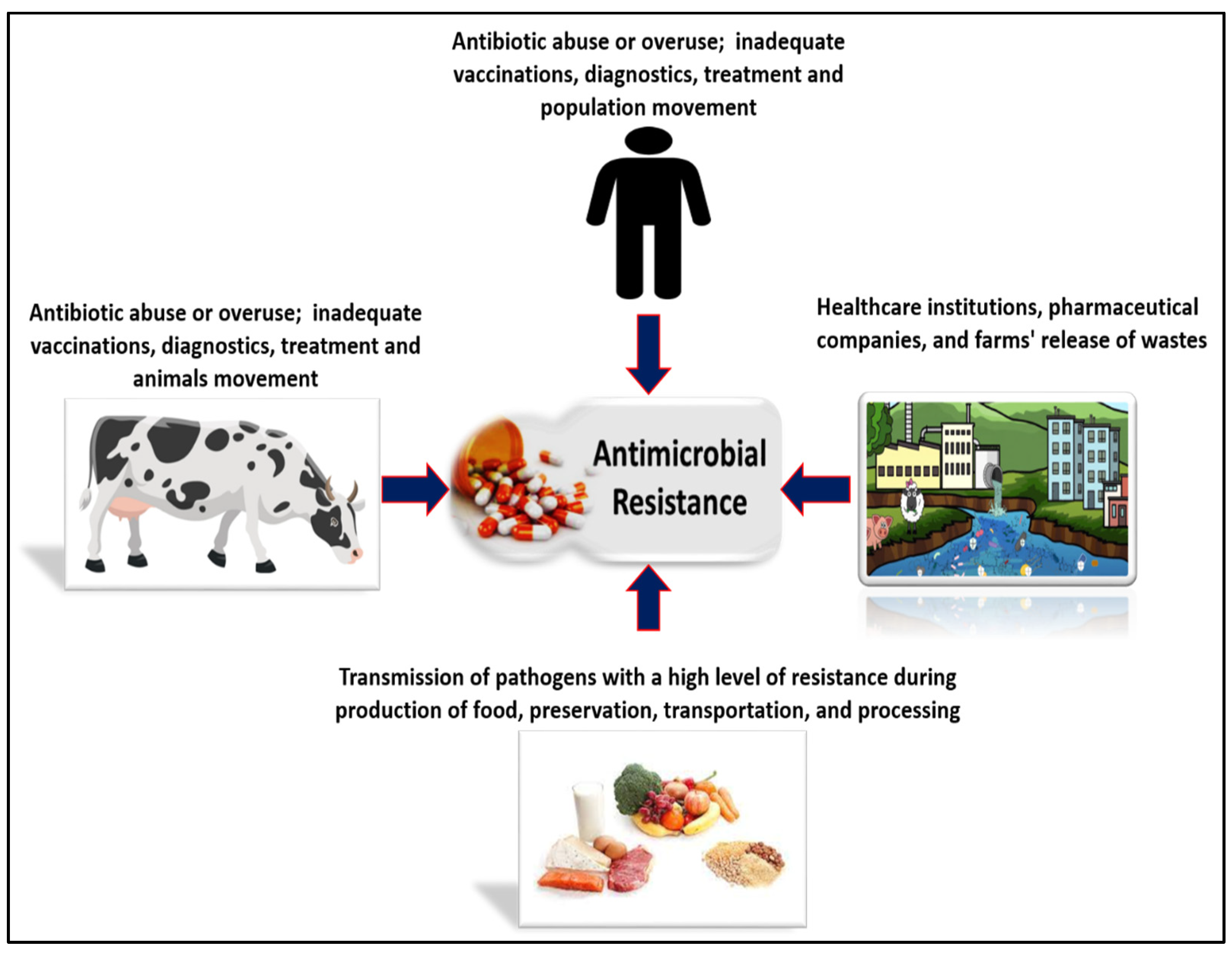
Vaccines, Free Full-Text

cdn.unenvironment.org/s3fs-public/inline-images/On

OMHHE Celebrates AANHPI Month - US FDA

Preparing for Medical Device Audits

Resist AMR, WHO

The Future of Antibiotics and Resistance
Related products