Solved P2.26 A vessel containing 2.00 mol of an ideal gas
5
(387)
Write Review
More
$ 6.00
In stock
Description

Solved 25- A mole of ideal gas with Cp = (7/2) R and Cv=

Heat 3

kinetic-theory-of-gases

Answered: At 45.0 °C, a 14.00 L vessel is filled…
The weight of 350ml of diatomic gas at 0°C and 2 atm pressure is 1 g. The weight of one atom is? - Quora

Solved Consider 2.00 mol of an ideal gas with a constant
How many moles of an ideal gas are contained in 8.2 L at -73 °C and 50.6 kPa? - Quora

Lab for January 13: the ideal-gas law
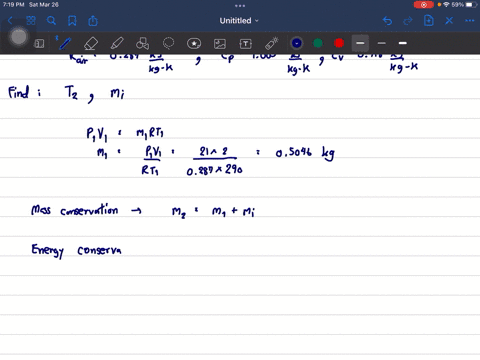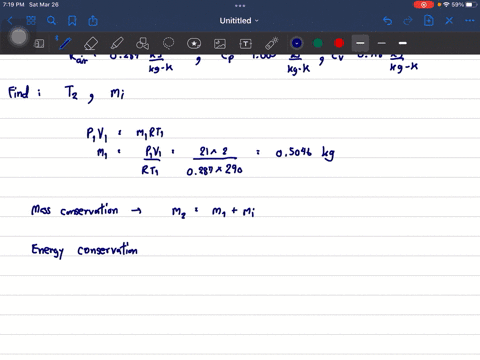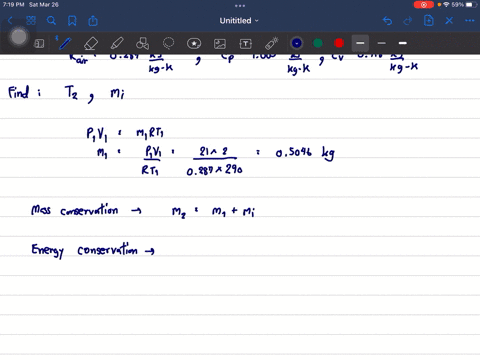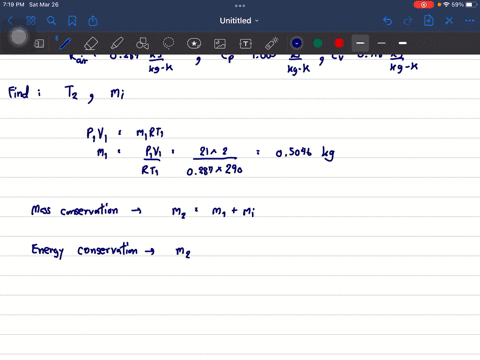
SOLVED: A vessel containing 2.00 mol of an ideal gas with P1 1.00 bar and Cp mR/2 is in thermal contact with a water bath. Treat the vessel, gas, and water bath

Solved Consider 2.00 mol of a monatomic ideal gas that is
Related products
You may also like